-
Thermal Properties of Matter
Heat, Temperature and its measurements
Introduction
William Thomson, 1st Baron Kelvin, (1824 –1907) was a British mathematical physicist, engineer, and outstanding leader in the physical sciences of the 19th century. He did important work in the mathematical analysis of electricity and thermodynamics, and did much to unify the emerging discipline of physics in its modern form. He is widely known for developing the Kelvin scale of absolute temperature measurement. The title Baron Kelvin was given in honor of his achievements, and named after the River Kelvin, which flowed past his university in Glasgow, Scotland.
Thermometers and the Celsius Temperature scale
Thermometers are devices that are used to measure temperatures. All thermometers are based on the principle that some physical properties of a system change as the system’s temperature changes. Some physical properties that change with temperature are
(1) the volume of a liquid
(2) the length of a solid
(3) the pressure of a gas at constant volume
(4) the volume of a gas at constant pressure and
(5) the electric resistance of a conductor.
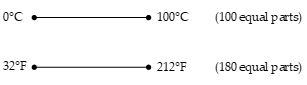
A common thermometer in everyday used consists of a mass of liquid, usually mercury or alcohol that expands in a glass capillary tube when heated. In this case the physical property is the change in volume of the liquid. Any temperature change is proportional to the change in length of the liquid column. The thermometer can be calibrated accordingly. On the Celsius temperature scale, a thermometer is usually calibrated between 0°C (called the ice point of water) and 100°C (called the steam point of water). Once the liquid levels in the thermometer have been established at these two points, the distance between the two points is divided into 100 equal segments to create the Celsius scale. Thus, each segment denotes a change in temperature of one Celsius degree (1°C). A practical problem in this type of thermometer is that readings may vary for two different liquids. When one thermometer reads a temperature, for example 40°C the other may indicate a slightly different value. This discrepancies between thermometers are especially large at temperatures far from the calibration points. To surmount this problem, we need a universal thermometer whose readings are independent of the substance used in it. The gas thermometer used in the next article meets this requirement.
The constant volume gas Thermometer and the absolute temperature scale
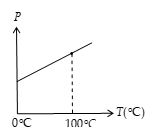
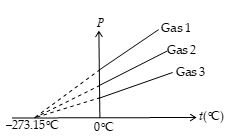
The physical property used by the constant volume gas thermometer is the change in pressure of a gas at constant volume.The pressure versus temperature graph for a typical gas taken with a constant volume is shown in figure. The two dots represent the two reference temperatures namely, the ice and steam points of water. The line connecting them serves as a calibration curve for unknown temperatures. Experiments show that the thermometer readings are nearly independent of the type of gas used as long as the gas pressure is low and the temperature is well above the point at which the gas liquefies.
If you extend the curves shown in figure toward negative temperatures, you find, in every case, that the pressure is zero when the temperatures is –273.15°C. This significant temperature is used as the basis for the absolute temperature scale, which sets –273.15°C as its zero point.
This temperature is often referred to as absolute zero. The size of a degree on the absolute temperature scale is identical to the size of a degree on the Celsius scale. Thus, the conversion between these temperatures is
TC = T – 273.15 ………….(i)
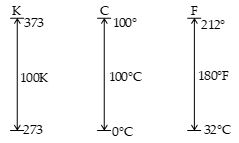
In 1954, by the International committee on weights and measures, the triple point of water was chosen as the reference temperature for this new scale. The triple point of water is the single combination of temperature and pressure at which liquid water, gaseous water and ice (solid water) coexist in equilibrium. This triple point occurs at a temperature of approximately 0.01°C and a pressure of 4.58 mm of mercury. On the new scale, which uses the unit kelvin, the temperature of water at the triple point was set at 273.16 kelvin, abbreviated as 273.16 K. (No degree sign is used with the unit Kelvin).
This new absolute temperature scale (also called the kelvin scale) employs the SI unit of absolute temperature, the Kelvin which is defined to be ‘‘\(\frac{1}{273.16}\)of the difference between absolute zero and the temperature of the triple point of water’’.
1)The Celsius, Fahrenheit and Kelvin Temperature Scales
Equation (i) shows the relation between the temperatures in Celsius scales and Kelvin scale. Because the size of a degree is the same on the two scales, a temperature difference of 10°C is equal to a temperature difference of 10 K. The two scales differ only in the choice of the zero point. The ice point temperature on the kelvin scale, 273.15 K, corresponds to 0.00°C and the Kelvin steam point 373.15 K, is equivalent to 100.00°C.
A common temperature scale is everyday use in US is the Fahrenheit scale. The ice point in this scale is 32°F and the steam point is 212°F. The distance between these two points are divided in 180 equal parts. The relation between Celsius scale and Fahrenheit scale is as derived below:
100 parts of Celsius scale = 180 parts of Fahrenheit scale
a) 1 part of Celsius scale \(=\frac{9}{5}\) parts of Fahrenheit scale
Hence,\({{T}_{F}}=32+\frac{9}{5}{{T}_{C}}\) …(ii)
Further, \(\Delta {{T}_{C}}=\Delta T=\frac{5}{9}\Delta {{T}_{F}}\) …(iii)
Conversion of Temperature from One Scale to Another
In order to convert temperature from one scale to another, following relation is used.
\({{\left( \frac{{{L}_{1}}}{{{L}_{2}}} \right)}^{2/3}}\)
\(\therefore \frac{3}{\left( \sqrt{2}+1 \right)}\)
Important Points in Thermometers
1. Different Thermometers Thermometric property: It is the property that can be used to measure the temperature. It is represented by any physical quantity such as length, volume, pressure and resistance etc., which varies linearly with a certain range of temperature. Let X denote the thermometric physical quantity and X0, X100 and Xt be its values at 0°C, 100°C and t°C respectively. Then,
\(t=\left( \frac{{{X}_{t}}-{{X}_{0}}}{{{X}_{100}}-{{X}_{0}}} \right)\times 100{}^\circ \text{C}\)
(i) Constant volume gas thermometer: The pressure of a gas at constant volume is the thermometric property. Therefore,
\(t=\left( \frac{{{P}_{t}}-{{P}_{0}}}{{{P}_{100}}-{{P}_{0}}} \right)\times 100{}^\circ \text{C}\)
(ii) Platinum resistance thermometer: The resistance of a platinum wire is the thermometric property. Hence,
\(t=\left( \frac{{{R}_{t}}-{{R}_{0}}}{{{R}_{100}}-{{R}_{0}}} \right)\times 100{}^\circ \text{C}\)
(iii) Mercury thermometer: In this thermometer the length of a mercury column from some fixed point is taken as thermometric property. Thus,
\(t=\left( \frac{{{l}_{t}}-{{l}_{0}}}{{{l}_{100}}-{{l}_{0}}} \right)\times 100{}^\circ \text{C}\)
2.Two other thermometers, commonly used are thermocouple thermometer and total radiation pyrometer.
3.Total radiation pyrometer is used to measure very high temperatures. When a body is at a high temperature, it glows brightly and the radiation emitted per second from unit area of the surface of the body is proportional to the fourth power of the absolute temperature of the body. If this radiation is measured by some device, the temperature of the body is calculated. This is the principle of a total radiation pyrometer. The main advantage of this thermometer is that the experimental body is not kept in contact with it. Hence, there is no definite higher limit of its temperature-range. It can measure temperature from 800°C to 3000°C–4000°C. However, it cannot be used to measure temperatures below 800°C because at low temperatures the emission of radiation is so poor that it cannot be measured directly.
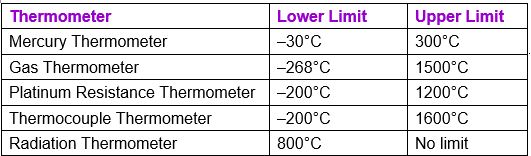
4.Ranges of different thermometers:
5.Reaumer’s Scale: Other than Celsius, Fahrenheit and Kelvin temperature scales Reaumer’s Scale was designed by Reaumer in 1730. The lower fixed point is 0°R representing melting point of ice. The upper fixed point is 80°R, which represents boiling point of water. The distance between the two fixed points is divided into 80 equal parts. Each part represents 1°R. If TC, TF and TR are temperature values of a body on Celsius scale, Fahrenheit scale and Reaumer scale respectively, then,
\(\frac{{{T}_{C}}-0}{100}=\frac{{{T}_{F}}-32}{180}=\frac{{{T}_{R}}-0}{80}\)
6. A substance is found to exist in three states solid, liquid and gas. For each substance there is a set of temperature and pressure at which all the three states may coexist. This is called triple point of that substance. For water, the values of pressure and temperature corresponding to triple point are 4.58 mm of Hg and 273.16°K.
Quantity of Heat
When a cold body is brought in contact with a hot body, the cold warms up and the hot body cools down as they approach thermal equilibrium. Fundamentally a transfer of energy takes place from one substance to the other. This type of energy transfer that takes place solely because of a temperature difference is called heat flow or heat transfer and energy transfer in this way is called heat.
Water can be warmed up by vigorous stirring with a paddle wheel. The paddle wheel adds energy to the water by doing work on it. The same temperature change can also be caused by putting the water in contact with some hotter body. Hence, this interaction must also involve an energy exchange. Before exploring the relation between heat and mechanical energy let us define a unit of quantity of heat.
‘‘One calorie (1 cal) is defined as the amount of heat required to raise the temperature of one gram of water from 14.5°C to 15.5°C.’’
Experiments have shown that 1 cal = 4.186 J
Similarly , 1 kcal = 1000 cal = 4186 J
The calorie is not a fundamental SI unit.
or \(\frac{1}{\sqrt{3}\,\,\left( \sqrt{2}-1 \right)}\)